Besides inclusion and large Ag (or WC) aggregations, the main defect inside AgWC material is the loose structure (see Fig.1), which can be easily destroyed by arc therefore increases the consumption of contact.

Figure 1. Fracture image of AgWC material with defects

Figure 2. Image of loose structure inside AgWC material.
Fig. 2 is an enlarged image for loose structure, where exposed WC particles are surrounded by non-continuous Ag network, indicates that Ag failed to infiltrate this area in the process. Actually, the nature of infiltration is the transport of liquid Ag in the product skeleton, thus we analyzed this transport process by scanning electron microscope (SEM) and provided certain methods to improve infiltration on that basis.
Ⅱ. Experiment
The basic process of AgWC material is: powder mixing, granulating, compacting, sintering, infiltrating and after-treatment. A JEOL JSM-6390A scanning electron microscopic was used to check the fracture morphology, and a SANS-CMT universal testing machine was used to measure the fracture resistance. Also of note is that contact materials are most likely to break where loose structure located under external force, for this reason, we recorded fracture resistance to represent the bond strength of contact material in this work.
Ⅲ. Results and discussion
1. The transport of liquid Ag in product skeleton
During infiltration, melted Ag, driven by the capillary force, infiltrates into the product skeleton and fills the pore network inside. Although the transport of liquid Ag starts from melting and end with solidification, experiments show that every process stage before infiltration has a significant influence on Ag transport, thus we observed the fracture morphology of product after each process stage to describe the whole process.
Mixed AgWC powder is compacted into skeleton with certain porosity during compaction. Fig. 3(a) shows the fracture of skeleton after die compaction, where Ag and WC particles distribute alternatively together with narrow pores, which form an internal network among the skeleton.
The skeleton would not be used for infiltration directly after compaction, but always has to be sintered under the protection of reducing atmosphere, which is called the ‘pre-sintering’. As shown in Fig. 3(b), the internal structure of the skeleton experienced remarkable changes during pre-sintering: the Ag particles sintered fiercely and wrapped around the surrounding WC particles. The tiny pores discharged during sintering aggregated into large pores simultaneously. Therefore the pre-sintering process is very similar to a primary infiltration, during which Ag particles infiltrated into structures around, resulted in the redistribution of pore network in skeleton.
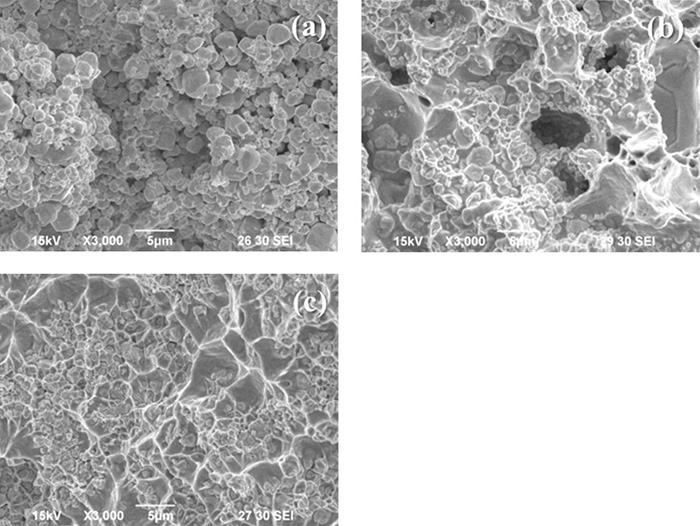
Figure 3. Fracture images of (a) AgWC skeleton after compaction, (b) AgWC skeleton after pre-sintering, and (c) AgWC product after infiltration.
During infiltration, the Ag plates beneath skeleton melted and infiltrated inside through the pore network formed during pre-sintering. As shown in Fig. 3(c), the highlight vein is tearing Ag, which completely wraps around the WC particles and fills the pore inside skeleton.
2. Methods to improve AgWC infiltration
Two methods are suggested here for a better infiltration of AgWC material based on the above analysis:
a)To enhance the transport dynamic of liquid Ag
The main driving force of infiltration is the capillary force, which can be analyzed in terms of interfacial energy. Take liquid Ag and WC particle for example: a lower interfacial energy derives a better wettability, thus results in a larger capillary force. Although the interfacial energy is mostly determined by the instinct properties of Ag and WC molecules, however, there are at least two methods to reduce the interfacial energy between liquid Ag and WC.
b)To use proper additives, for example Cu and Ni.
For one thing, these additives dissolve in Ag in a certain degree, for another, the interfacial energy between WC and additives is much lower than that of Ag. As a result, additives play the role of surfactant and reduce the overall interfacial energy. Fig. 4(a) and Fig. 4(b) show the fracture of AgWC materials with and without additives. It can be concluded that additives could significantly improve infiltration thus reduce defects inside the material.

Figure 4. Fracture images of AgWC materials (a) with additives and (b) without additives
To increase infiltration temperature. Under fixed pressure, a higher temperature results in a higher average kinetic energy of molecule which leads to a larger molecular separation, means a smaller molecular attraction and finally a lower interfacial energy. Therefore increasing temperature is the most efficient method to improve infiltration. Fig. 5 presents the fracture resistance data of products infiltrated at different temperatures. It can be seen that the fracture resistance increases with increasing infiltration temperature, which demonstrates that the increasing of temperature improves the infiltration process.
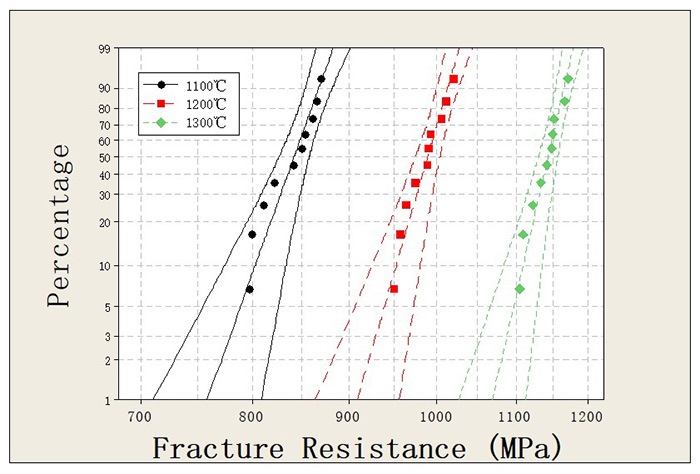
Figure 5. Fracture resistance data of AgWC materials infiltrated at 1100℃ (black curve), 1200℃ (red curve) and 1300℃ (green curve).
a) To broaden the ‘transport corridor’ of liquid Ag
An excellent infiltration process needs Ag to infiltrate throughout skeleton thoroughly. Any poor-infiltrated area will become defect and decreases the anti-erosion ability of the product. As discussed above, Ag infiltrates inside skeleton through the pore network formed in the process stage of compaction. However, for one thing, these pores are too narrow for liquid Ag to flow, which will definitely extend infiltration time, for another, certain amount of tiny pores will get stuck inside and turn to defect finally. Hence the pore network should be broadened for a better transport of liquid Ag.
b)To increase pre-sintering temperature.
As discussed above, the pore network inside skeleton redistributes and the pore size increases due to the primary infiltration in pre-sintering process which infers that pre-sintering is an effective way to broaden the pore channel. Because pre-sintering is similar to infiltration, so a proper increase in temperature is likely to improve the sintering degree of skeleton thus induce a broader pore network. It could be seen in Fig. 6 that the pore size in skeleton increases with the increasing sintering temperature. The fracture resistance of product also increases with the pre-sintering temperature (see Fig. 7), implies the improvement to infiltration process.

Figure 6. Fracture images of AgWC skeleton sintered at (a) 1000℃, (b) 1100℃ and (c) 1200℃.
c)To add pore-forming agent.
Generally, the pore-forming agent is added in powder mixing process and removed after die-compaction. The larger the particle size of the agent, the better result it brings. The pore-forming agent occupies the space of original pores in skeleton during compaction and releases much larger pores when removed. These large pores are filled by Ag after infiltration, as shown in Fig. 8 (the smooth islands in the structure). Moreover, those spaces occupied by Ag particles in skeleton turns to pore channels during infiltration as a result of Ag melting, which actually offers an external transport corridor for liquid Ag, for this reason, increasing the size of Ag particle is equivalent to adding pore-forming agent to a certain extent.
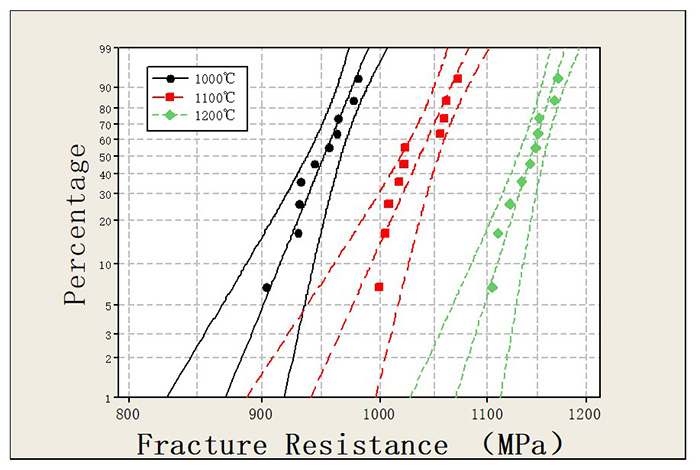
Figure 7. Fracture resistance data of AgWC material produced with pre-sintering temperature of 1000℃ (black curve), 1100℃ (red curve) and 1200℃ (green curve).

Figure 8. Fracture image of AgWC material manufactured with the help of pore-forming agent.
Ⅳ. Conclusion
This work observed the structure morphology of AgWC material after each process stage by SEM, and analyzed the mechanism of the liquid Ag transport during infiltration. Based on the discussion, we proposed two methods to improve the infiltration of AgWC material: first is to reduce the interfacial energy between Ag and WC, which could be realized by adding proper additives like Cu and Ni, and increasing the infiltration temperature; second is to broaden the infiltration corridor for liquid Ag, which could be achieved by increasing the pre-sintering temperature and adding pore-forming agent. The result could also be applied to other materials manufactured through infiltration.







