conducted experiments to compare the performance of silver tungsten (AgW) and silver tungsten carbide (AgWC) in various corrosive environments. The findings offer valuable insights for selecting and optimizing contact materials.
Why Study the Corrosion Resistance of AgW and AgWC?
AgW and AgWC contact materials are widely used in circuit breakers for their excellent electrical conductivity and resistance to arc burns. However:
- Challenges of AgW contacts: Tungsten is prone to oxidation, especially in high-temperature and high-humidity environments. This oxidation forms a non-conductive tungstate film, which increases contact resistance.
Advantages of AgWC contacts: Tungsten carbide (WC) is chemically more stable than tungsten. Its corrosion potential is close to that of silver, which theoretically enhances its corrosion resistance.
The ability of AgW and AgWC to withstand various corrosive environments was systematically evaluated. This was done using both a high-low temperature alternating damp heat test and a salt spray test.
Experimental Methods and Conditions
Preparation of Samples
AgW and AgWC contact materials were prepared using the infiltration method. AgW samples were divided into three groups, with 0.5%, 1.5%, and 2.5% corrosion-resistant additives. These groups were labeled as AgW-1, AgW-2, and AgW-3. AgWC samples were added with 1.5% additives as a control.
| Material |
Ingredient Content,wt% |
| Ag |
W/WC |
T(Additive) |
| AgW-1 |
35 |
Balance |
0-0.5 |
| AgW-2 |
50 |
Balance |
1-1.5 |
| AgW-3 |
35 |
Balance |
2-2.5 |
| AgWC |
60 |
Balance |
1-1.5 |
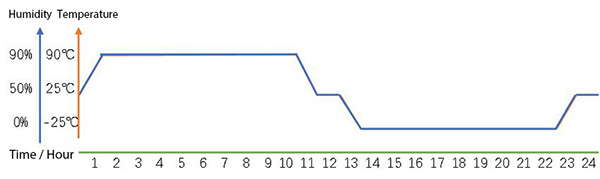
High-low Temperature Alternating Damp Heat Test
14 cycles in a damp heat test chamber:
- Temperature range: -25°C to 90°C alternating cycles;
- Humidity range: 0% to 90%;
- Simulates corrosive environments under high humidity condensing conditions.
Salt Spray Test
Salt spray corrosion test according to GB/T 6458 standard:
- Temperature: 35℃
- Concentration of sodium chloride solution: 5%
- Continuous spray time: 240 hours
Experimental Results and Analysis
High-low temperature alternating damp heat test
- AgW contact: AgW-1, without additives, oxidizes the fastest. As the additive content increases (AgW-2, AgW-3), the oxidation rate gradually decreases. However, signs of oxidation are still observed after 14 cycles.
- AgWC contact: No oxidation phenomenon was found after 14 cycles, and the surface condition was intact.
Salt Spray Test
All contact samples showed no significant oxidation after 240 hours of testing.
Corrosion Resistance Conclusion
- In hot and humid environments, AgWC contacts exhibit significantly better corrosion resistance than AgW contacts.
- In salt spray environments, AgW and AgWC contacts are close in corrosion resistance.
Degree of Oxidation of the Product Appearance After the High-low Temperature Alternating Damp Heat Test and Salt Spray Test (Visual Assessment)
| Material |
Ingredient Content,wt% |
High-low Temperature Alternating Damp Heat Test |
Salt Spray Test |
| Ag |
W/WC |
T(Additive) |
Cycle 6 |
Cycle 14 |
72 Hours |
240Hours |
| AgW-1 |
35 |
Balance |
0-0.5 |
Heavily oxidized |
Very heavily oxidized |
No oxidation observed |
No oxidation observed |
| AgW-2 |
50 |
Balance |
1-1.5 |
No oxidation observed |
Heavily oxidized |
No oxidation observed |
No oxidation observed |
| AgW-3 |
35 |
Balance |
2-2.5 |
No oxidation observed |
Slightly oxidised |
No oxidation observed |
No oxidation observed |
| AgWC |
60 |
Balance |
1-1.5 |
No oxidation observed |
No oxidation observed |
No oxidation observed |
No oxidation observed |
Photographs of Product Appearance After the Test
| Material |
After 14 Cycles of High-low Temperature Alternating Damp Heat Test |
After 240 Hours of Salt Spray Test |
| AgW-1 |

|

|
| AgW-2 |

|

|
| AgW-3 |

|

|
| AgWC |

|

|
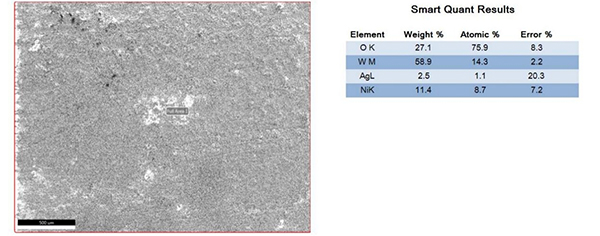
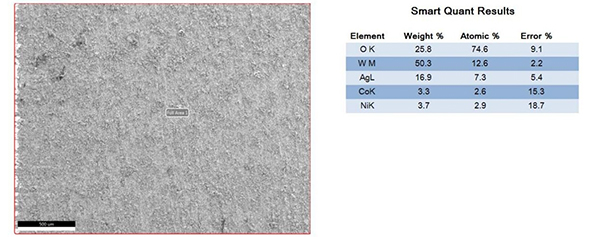
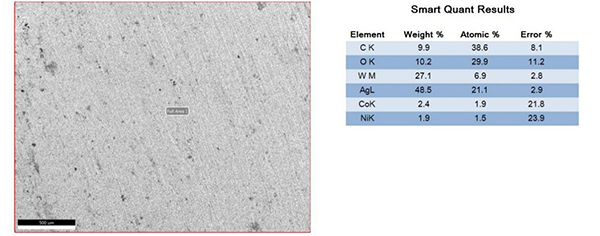
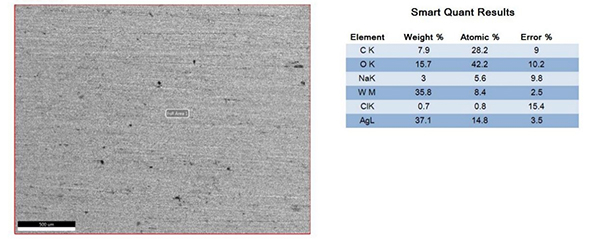
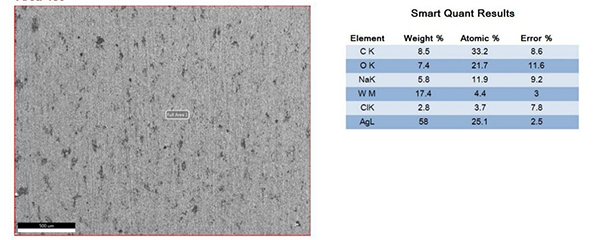
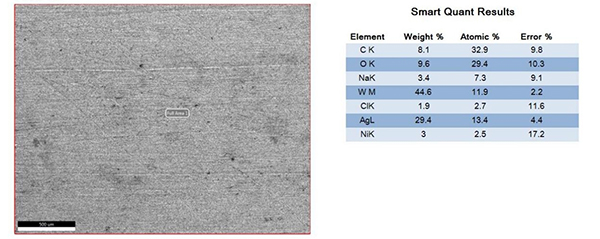
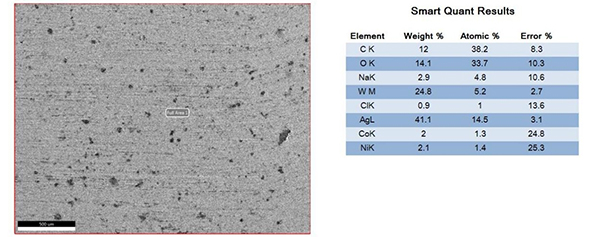
The surfaces of the electrical contacts after the test were examined using a scanning electron microscope to analyze their degree of oxidation (judged by the changes in the surface Ag, O, and W content). Figures 2 to 5 display the surface morphology of the four types of electrical contacts. These images were captured after the 14th cycle of the high-low temperature alternating damp heat test.
Figure 2. Surface Morphology and Electron Microscope Scanning Results After the 14th Cycle of AgW-1 Alternating Damp Heat Test
Figure3. Surface Morphology and Electron Microscope Scanning Results After the 14th Cycle of AgW-2 Alternating Damp Heat Test
Figure4. Surface Morphology and Electron Microscope Scanning Results After the 14th Cycle of AgW-3 Alternating Damp Heat Test
Figure5. Surface Morphology and Electron Microscope Scanning Results After the 14th Cycle of AgWC Alternating Damp Heat Test
The data in the table and the scanning electron microscope analyses in Figures 2 to 5 indicate that, in the high-low temperature alternating damp heat test, the oxidation resistance of the AgW product improves as the content of additives increases. On the other hand, the AgWC product remains unaffected by the alternating humidity and heat. After 14 cycles, the surface of the AgWC product remains unchanged.
Figures 6 to 9 show the surface morphology of the four types of electrical contacts after 240 hours of salt spray test.
Figure 6. Surface Morphology and Electron Microscope Scanning Results of AgW-1 After 240 Hours of Salt Spray Test
Figure 7. Surface Morphology and Electron Microscope Scanning Results of AgW-2 After 240 Hours of Salt Spray Test
Figure 8. Surface Morphology and Electron Microscope Scanning Results of AgW-3 After 240 Hours of Salt Spray Test
Figure 9. Surface Morphology and Electron Microscope Scanning Results of AgWC After 240 Hours of Salt Spray Test
The tabular data and SEM analyses in Figures 6 to 9 show that no significant oxidation occurred on the AgW and AgWC electrical contacts. This was observed after the 240-hour salt spray test.
Analysis of Corrosion Resistance Mechanism
The nature of metal corrosion is a redox reaction. Under high-low temperature alternating damp heat environment:
-Disadvantage of AgW contact: Higher tungsten activity makes it more prone to reacting with the electrolyte. This reaction generates a tungstate film, which leads to increased corrosion.
- Advantage of AgWC contacts: Lower tungsten carbide activity, closer to the electrochemical properties of silver, significantly lower corrosion rate.
Under the salt spray environment, the corrosion rate is more limited by the temperature, and the oxidation process on the surface of AgW and AgWC contacts is inhibited, so the performance is similar.
Conclusions and Practical Applications
AgWC contacts outperform AgW contacts in hot and humid environments. They are recommended for use in high-temperature and high-humidity conditions. In salt spray environments, both types of contacts perform equally well. Materials can be selected based on cost and other performance requirements.
AgWC contacts are suitable for harsh environments, such as marine equipment and high-humidity factories. On the other hand, AgW contacts remain cost-effective in normal environments. Optimizing additive compositions can further enhance their corrosion resistance.
This study not only reveals the corrosion behavior of AgW and AgWC in different environments but also provides a scientific basis for the design and selection of electrical contact materials. As the performance requirements for contact materials increase, high-performance materials like AgWC will become more important. They will play a key role in a wider range of fields. For more information about the AgCdO and related solutions or products, please visit www.fudarworld.com or feel free to contact us.